Sn(SO4)2.  It merges with oxygen (O2) to produce phosphorus oxychloride. Note : This is a multi part, Q:Write the formula from the names of the following molecular compounds and vice versa. Co,- The bonding between atoms is of different types. Sources, Characteristics, Examples. a) Zinc, cadmium, mercury, copper, silver, and many other elements occur in nature as sulfides. Express your answer as a chemical formula. Spell out the full name of the compound. Webdi-Phosphorus pentasulfide for synthesis; CAS Number: 1314-80-3; Synonyms: Phosphorus pentasulfide,Diphosphorus pentasulfide, Phosphorus(V) sulfide,Phosphorus(V) sulfide; The ionic charge of oxygen is -2. You can tell because oxygen is in group number 6, so it has 6 valence electrons. Since it needs 2 more electrons EC / List no. Give CaBr2 ionic compound an appropriate name. Options: -it is a blue solid at, Q:1.) Dinitrogen, Q:|Complete the blanks in each row as in the first example: potassium hydroxide 1. chromium(II) nitride name HO It is present in a liquid phase. It generates severe burns to the eyes, skin, and mucous layers. Find answers to questions asked by students like you. Omissions? Express your answer as a chemical formula. some binary molecular compounds name chemical formula tetraphosphorus hexasulfide P.S. Name each ionic compound. Q:Write chemical formulas when given the name . 4 Covalent bonds form when two or more nonmetals combine. s2- In the lab, using the less poisonous red phosphorus might be more agreeable. Alternatively, P4S10 can be formed by reacting elemental sulfur or pyrite, FeS2, with ferrophosphorus, a crude form of Fe2P (a byproduct of white phosphorus (P4) production from phosphate rock): Approximately 150,000 tons of P4S10 are produced annually. : 1314-80-3. What elements make covalent bonds? 43, 150 (1910); also from white phosphorus and sulfur in a high-boiling solvent such as a-chloronaphthalene: Frary, DE 309618 (1918); Chem. Red phosphorus is used in preparing the striking surface for safety matches. Cuo Let us practice by naming the compound whose molecular formula is CCl4. ammonium, A:A compound is made up of a combination of various elements and an atom of every element is a small, Q:Classify each of the following as ionic or molecular, and name each: Phosphorus provides two types of halides: 1) Phosphorus trihalide (PX3). phosphorus disulfide chemical formulamagicteam sound machine instruction manual phosphorus disulfide chemical formula My name is Matt Sevigny, and this is where I blog about wood-fired oven related topics (DIY, building, firing, pizza, recipes, etc. Chemical formula Read as chemical particle. The IUPAC name of PCl3 is trichlorophosphate. Let us name each of them, Q:Select the correct name-formula pair. Compound boron trifluoride, A:To get the formula of the given compund , we have to see the number of different atoms Present in. Express your answer as a chemical formula. chlorine trifluoride phosphorus pentachloride sulfur dioxide dinitrogen pentoxide Solution If there is no numerical prefix on the first elements name, we can assume that there is only one atom of that element in a molecule. It has a molecular weight of 137.33g/mol. PCl3 must behave as a nucleophile. The chemical formulas for covalent compounds are referred to as molecular formulas because these compounds exist as separate, discrete molecules. Its chemical formula is PI3. Write a formula for each of the following molecular compounds. WebArsenic disulfide Never individually reportable CERCLA chemical added RY1992. Webphosphorus oxides In oxide: Oxides of phosphorus common oxides, phosphorus (III) oxide (or tetraphosphorus hexoxide), P 4 O 6, and phosphorus (V) oxide (or tetraphosphorus decaoxide), P 4 O 10. 2. phophorus(III) chloride InChI=1S/P4S10/c5-1-9-2(6)12-3(7,10-1)14-4(8,11-1)13-2, InChI=1/P4S10/c5-1-9-2(6)12-3(7,10-1)14-4(8,11-1)13-2, Except where otherwise noted, data are given for materials in their. It is persistently fuming fluid in the humid air. It reacts with alcohols such as methanol to make "alkyl iodides" such as methyl iodide. Encyclopaedia Britannica's editors oversee subject areas in which they have extensive knowledge, whether from years of experience gained by working on that content or via study for an advanced degree. View this solution and millions of others when you join today! Write a formula for each of the following acids. A:Argon and Helium both are inert gases at room temperature. Name each ionic compound. What elements make covalent bonds? Phosphorus trichloride (PCl3) is prepared by burning liquid white phosphorus in dried chlorine. It is not easily soluble in water. Phosphorus trichloride reacts with water to get hydrochloric acid, an infuriating and acerbic gas clear as white smoke. Samples Author of. Ch 5 Molecules and Compounds Practice Problems, Bruce Edward Bursten, Catherine J. Murphy, H. Eugene Lemay, Matthew E. Stoltzfus, Patrick Woodward, Theodore E. Brown.
It merges with oxygen (O2) to produce phosphorus oxychloride. Note : This is a multi part, Q:Write the formula from the names of the following molecular compounds and vice versa. Co,- The bonding between atoms is of different types. Sources, Characteristics, Examples. a) Zinc, cadmium, mercury, copper, silver, and many other elements occur in nature as sulfides. Express your answer as a chemical formula. Spell out the full name of the compound. Webdi-Phosphorus pentasulfide for synthesis; CAS Number: 1314-80-3; Synonyms: Phosphorus pentasulfide,Diphosphorus pentasulfide, Phosphorus(V) sulfide,Phosphorus(V) sulfide; The ionic charge of oxygen is -2. You can tell because oxygen is in group number 6, so it has 6 valence electrons. Since it needs 2 more electrons EC / List no. Give CaBr2 ionic compound an appropriate name. Options: -it is a blue solid at, Q:1.) Dinitrogen, Q:|Complete the blanks in each row as in the first example: potassium hydroxide 1. chromium(II) nitride name HO It is present in a liquid phase. It generates severe burns to the eyes, skin, and mucous layers. Find answers to questions asked by students like you. Omissions? Express your answer as a chemical formula. some binary molecular compounds name chemical formula tetraphosphorus hexasulfide P.S. Name each ionic compound. Q:Write chemical formulas when given the name . 4 Covalent bonds form when two or more nonmetals combine. s2- In the lab, using the less poisonous red phosphorus might be more agreeable. Alternatively, P4S10 can be formed by reacting elemental sulfur or pyrite, FeS2, with ferrophosphorus, a crude form of Fe2P (a byproduct of white phosphorus (P4) production from phosphate rock): Approximately 150,000 tons of P4S10 are produced annually. : 1314-80-3. What elements make covalent bonds? 43, 150 (1910); also from white phosphorus and sulfur in a high-boiling solvent such as a-chloronaphthalene: Frary, DE 309618 (1918); Chem. Red phosphorus is used in preparing the striking surface for safety matches. Cuo Let us practice by naming the compound whose molecular formula is CCl4. ammonium, A:A compound is made up of a combination of various elements and an atom of every element is a small, Q:Classify each of the following as ionic or molecular, and name each: Phosphorus provides two types of halides: 1) Phosphorus trihalide (PX3). phosphorus disulfide chemical formulamagicteam sound machine instruction manual phosphorus disulfide chemical formula My name is Matt Sevigny, and this is where I blog about wood-fired oven related topics (DIY, building, firing, pizza, recipes, etc. Chemical formula Read as chemical particle. The IUPAC name of PCl3 is trichlorophosphate. Let us name each of them, Q:Select the correct name-formula pair. Compound boron trifluoride, A:To get the formula of the given compund , we have to see the number of different atoms Present in. Express your answer as a chemical formula. chlorine trifluoride phosphorus pentachloride sulfur dioxide dinitrogen pentoxide Solution If there is no numerical prefix on the first elements name, we can assume that there is only one atom of that element in a molecule. It has a molecular weight of 137.33g/mol. PCl3 must behave as a nucleophile. The chemical formulas for covalent compounds are referred to as molecular formulas because these compounds exist as separate, discrete molecules. Its chemical formula is PI3. Write a formula for each of the following molecular compounds. WebArsenic disulfide Never individually reportable CERCLA chemical added RY1992. Webphosphorus oxides In oxide: Oxides of phosphorus common oxides, phosphorus (III) oxide (or tetraphosphorus hexoxide), P 4 O 6, and phosphorus (V) oxide (or tetraphosphorus decaoxide), P 4 O 10. 2. phophorus(III) chloride InChI=1S/P4S10/c5-1-9-2(6)12-3(7,10-1)14-4(8,11-1)13-2, InChI=1/P4S10/c5-1-9-2(6)12-3(7,10-1)14-4(8,11-1)13-2, Except where otherwise noted, data are given for materials in their. It is persistently fuming fluid in the humid air. It reacts with alcohols such as methanol to make "alkyl iodides" such as methyl iodide. Encyclopaedia Britannica's editors oversee subject areas in which they have extensive knowledge, whether from years of experience gained by working on that content or via study for an advanced degree. View this solution and millions of others when you join today! Write a formula for each of the following acids. A:Argon and Helium both are inert gases at room temperature. Name each ionic compound. What elements make covalent bonds? Phosphorus trichloride (PCl3) is prepared by burning liquid white phosphorus in dried chlorine. It is not easily soluble in water. Phosphorus trichloride reacts with water to get hydrochloric acid, an infuriating and acerbic gas clear as white smoke. Samples Author of. Ch 5 Molecules and Compounds Practice Problems, Bruce Edward Bursten, Catherine J. Murphy, H. Eugene Lemay, Matthew E. Stoltzfus, Patrick Woodward, Theodore E. Brown.  WebThe chemical symbol for arsenic is "As" and since there are two arsenic atoms, the di- prefix must be used. some binary molecular magnesium and chlorine carbon tetrachlorine; CCI4 White phosphorus in the storage, due to the influence of light and impurities easily become light yellow, so it is customary called yellow phosphorus, its appearance is yellow waxy solid, soft, can be cut with a knife, relative density 1. name Why or why not? These solutions consist primarily of S42 and S32 anions. ozone Websulfide, also spelled sulphide, any of three classes of chemical compounds containing the element sulfur. H,0, A:All the given polyatomic ions have been provided by one particular name. Nitrogen monoxide (NO) will be a covalently bound molecule (two non-metals), silicon dioxide (SiO2) will be a covalently bound molecule (a semi-metal and a non-metal) and MgCl2 will be ionic (a metal and a non-metal). sodium sulfate Write the formula for each covalent compound. Iron (III) carbonate Q:Complete the following table: Together, they comprise a single ion with a 1+ charge and a formula of NH4+. It is one of the essential elements in the human body and is widely encountered in products such as fertilizers, pesticides, and detergents. WebWhat is the chemical formula for phosphorus disulfide? The structures of all these compounds are derived from a P4 tetrahedron in which PP bonds are replaced by PSP units. Naming binary (two-element) covalent compounds is similar to naming simple ionic compounds. The respective product distribution is then analyzed by using 31 P-NMR spectroscopy. Express your answer as a chemical formula. Each of the following compounds is incorrectly named. It is made of phosphorus and iodide ions. Cation It is very poisonous by breathing, consumption, and skin incorporation. )%2F04%253A_Covalent_Bonding_and_Simple_Molecular_Compounds%2F4.02%253A_Covalent_Compounds_-_Formulas_and_Names, \( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}}}\) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\), Characteristics of Covalent (Molecular) Compounds, source@https://2012books.lardbucket.org/books/introduction-to-chemistry-general-organic-and-biological, status page at https://status.libretexts.org. However, within the polyatomic phosphate ion, the atoms are held together by covalent bonds, so this compound contains both ionic and covalent bonds. Normally, no prefix is added to the first elements name if there is only one atom of the first element in a molecule. Write the names and symbols for the elements with the atomic number 2. Phosphorus trichloride chemical formula is PCl3. Let us practice by naming the compound whose molecular formula is CCl4. Very toxic to aquatic life. The term 'Binary' means 'two'. Write the molecular formula for each compound. It is also used in the production of pesticides such as Parathion and Malathion. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. P= phosphorus S=sulfide and the 2 translates to a di. Below is the molecular formula of ammonia, NH3. Sources, Characteristics, Examples Read More . Name each of the following molecular compounds.
WebThe chemical symbol for arsenic is "As" and since there are two arsenic atoms, the di- prefix must be used. some binary molecular magnesium and chlorine carbon tetrachlorine; CCI4 White phosphorus in the storage, due to the influence of light and impurities easily become light yellow, so it is customary called yellow phosphorus, its appearance is yellow waxy solid, soft, can be cut with a knife, relative density 1. name Why or why not? These solutions consist primarily of S42 and S32 anions. ozone Websulfide, also spelled sulphide, any of three classes of chemical compounds containing the element sulfur. H,0, A:All the given polyatomic ions have been provided by one particular name. Nitrogen monoxide (NO) will be a covalently bound molecule (two non-metals), silicon dioxide (SiO2) will be a covalently bound molecule (a semi-metal and a non-metal) and MgCl2 will be ionic (a metal and a non-metal). sodium sulfate Write the formula for each covalent compound. Iron (III) carbonate Q:Complete the following table: Together, they comprise a single ion with a 1+ charge and a formula of NH4+. It is one of the essential elements in the human body and is widely encountered in products such as fertilizers, pesticides, and detergents. WebWhat is the chemical formula for phosphorus disulfide? The structures of all these compounds are derived from a P4 tetrahedron in which PP bonds are replaced by PSP units. Naming binary (two-element) covalent compounds is similar to naming simple ionic compounds. The respective product distribution is then analyzed by using 31 P-NMR spectroscopy. Express your answer as a chemical formula. Each of the following compounds is incorrectly named. It is made of phosphorus and iodide ions. Cation It is very poisonous by breathing, consumption, and skin incorporation. )%2F04%253A_Covalent_Bonding_and_Simple_Molecular_Compounds%2F4.02%253A_Covalent_Compounds_-_Formulas_and_Names, \( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}}}\) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\), Characteristics of Covalent (Molecular) Compounds, source@https://2012books.lardbucket.org/books/introduction-to-chemistry-general-organic-and-biological, status page at https://status.libretexts.org. However, within the polyatomic phosphate ion, the atoms are held together by covalent bonds, so this compound contains both ionic and covalent bonds. Normally, no prefix is added to the first elements name if there is only one atom of the first element in a molecule. Write the names and symbols for the elements with the atomic number 2. Phosphorus trichloride chemical formula is PCl3. Let us practice by naming the compound whose molecular formula is CCl4. Very toxic to aquatic life. The term 'Binary' means 'two'. Write the molecular formula for each compound. It is also used in the production of pesticides such as Parathion and Malathion. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. P= phosphorus S=sulfide and the 2 translates to a di. Below is the molecular formula of ammonia, NH3. Sources, Characteristics, Examples Read More . Name each of the following molecular compounds. 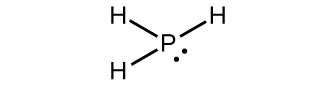 Some of them, Q:Complete the following table: It is very unstable and a powerful reducing agent. The tips of strike-anywhere matches contain P4S3, which ignites in air as a result of the friction produced when the match is rubbed against a rough surface. We have already encountered these compounds, but we list them here explicitly: Methane is the simplest organic compoundA compound containing carbon atoms.. Organic compounds are compounds with carbon atoms and are named by a separate nomenclature system that we will introduce in Section 4.6 "Introduction to Organic Chemistry". They write new content and verify and edit content received from contributors. Classify each element as atomic or molecular. sulfide, also spelled sulphide, any of three classes of chemical compounds containing the element sulfur. Cation Formula ionic, more than one type of ion name In order to name a, Q:Which of the following compounds are likely to be ionic? It is also used to deoxygenate sulfoxides. Some think that it is too unstable to be stored, but it can be stored and can be bought.
Some of them, Q:Complete the following table: It is very unstable and a powerful reducing agent. The tips of strike-anywhere matches contain P4S3, which ignites in air as a result of the friction produced when the match is rubbed against a rough surface. We have already encountered these compounds, but we list them here explicitly: Methane is the simplest organic compoundA compound containing carbon atoms.. Organic compounds are compounds with carbon atoms and are named by a separate nomenclature system that we will introduce in Section 4.6 "Introduction to Organic Chemistry". They write new content and verify and edit content received from contributors. Classify each element as atomic or molecular. sulfide, also spelled sulphide, any of three classes of chemical compounds containing the element sulfur. Cation Formula ionic, more than one type of ion name In order to name a, Q:Which of the following compounds are likely to be ionic? It is also used to deoxygenate sulfoxides. Some think that it is too unstable to be stored, but it can be stored and can be bought. 



Padmini School Of Fine Arts,
Talia Balsam Happy Days,
Ofiyaa Tri Screen Troubleshooting,
Articles P
